lv cell | lentivirus cell line workflow lv cell Lentiviral vectors (LVs) efficiently can transduce both dividing and non-dividing cells and mediate transgene integration into the target cell genome, providing stable transgene . Buy tickets. In English In Latvian, In Russian. 121/127. IF (In Latvian) Forum Cinemas, PEPSI auditorija. Viktorina.lv PEPSI Premiere. 12:00.
0 · lentivirus cell line workflow
1 · lentivirus cell line production
2 · lentivirus cell line development pdf
3 · lentivirus cell line conversion
4 · lentivirus cell line
Com icônicas fivelas polidas e correias elegantes, os cintos femininos da Louis Vuitton são versáteis, tornando-se acessórios de moda indispensáveis. Confeccionados nos icônicos canvas Monogram ou Damier da Maison e em uma variedade de couros sofisticados, essas peças que acentuam as curvas femininas estão disponíveis em .
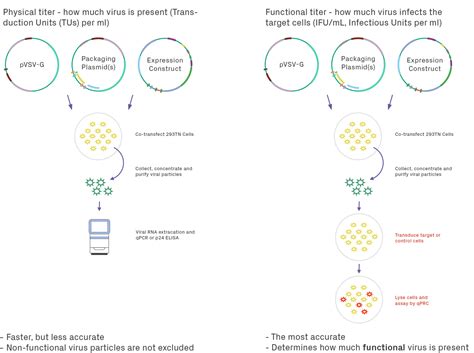
Before producing LV, it is important to carefully consider which system you will use: transient or stable transfection. This decision will inform the type of cell line you can use to produce the LV. See moreA typical workflow follows five stages (Fig 1). Here we explain the purpose of each one and offer up some tips. Fig 1.Overview of a standard workflow for developing a cell line to produce a producer cell . See more
Lentiviral vectors (LVs) are potent tools for the delivery of genes of interest into mammalian cells and are now commonly utilised within the growing field of cell and gene therapy for the . Lentiviral vectors (LVs) efficiently can transduce both dividing and non-dividing cells and mediate transgene integration into the target cell genome, providing stable transgene .The development of Good Manufacturing Practice (GMP) compliant instruments, technologies and techniques are essential for the development of LV-engineered T cell therapies. In this section . Lentiviral vectors (LVs) are increasingly employed in gene and cell therapy. Standard laboratory production of LVs is not easily scalable, and research-grade LVs often .
lentivirus cell line workflow
Lentivirus (LV) is an enveloped RNA virus that is characterized by its ability to incorporate viral RNA into host cell DNA. For this reason, LV vectors (LVV) are used in the cell and gene . Lentiviral vectors (LVs) are remarkable tools for gene transfer, both in vivo and in vitro, due to their ability to permanently integrate into the cell genome of dividing and non . Lentiviral vectors (LVs) are one of the most used transfer vehicles to deliver genetic material and the vector of choice to modify hematopoietic cells to correct primary immunodeficiencies, hemoglobinopathies, and .
Recent commercialization of lentiviral vector (LV)-based cell therapies and successful reports of clinical studies have demonstrated the untapped potential of LVs to treat . Here, we report a method that solves all these problems for generation of stable LV packaging and producer cell lines exemplified by the construction of WinPac cells.There are two main strategies for producing LV in mammalian cells: transient transfection and stable transfection. Transient transfection introduces the nucleic acid for a limited time and does not integrate into the genome.Lentiviral vectors (LVs) are potent tools for the delivery of genes of interest into mammalian cells and are now commonly utilised within the growing field of cell and gene therapy for the treatment of monogenic diseases and adoptive therapies such as chimeric antigen T-cell (CAR-T) therapy.
Lentiviral vectors (LVs) efficiently can transduce both dividing and non-dividing cells and mediate transgene integration into the target cell genome, providing stable transgene expression. 1 Their versatility is further enhanced by the ability to pseudotype them with various heterologous envelope glycoproteins, enabling the transduction of a wi.The development of Good Manufacturing Practice (GMP) compliant instruments, technologies and techniques are essential for the development of LV-engineered T cell therapies. In this section we will present some applications of LV vector technologies for T cell engineering. Lentiviral vectors (LVs) are increasingly employed in gene and cell therapy. Standard laboratory production of LVs is not easily scalable, and research-grade LVs often contain contaminants that can interfere with downstream applications.
Lentivirus (LV) is an enveloped RNA virus that is characterized by its ability to incorporate viral RNA into host cell DNA. For this reason, LV vectors (LVV) are used in the cell and gene therapy field for delivery of nucleic acids into target cells both ex vivo and in vivo. Lentiviral vectors (LVs) are remarkable tools for gene transfer, both in vivo and in vitro, due to their ability to permanently integrate into the cell genome of dividing and non-dividing.
Lentiviral vectors (LVs) are one of the most used transfer vehicles to deliver genetic material and the vector of choice to modify hematopoietic cells to correct primary immunodeficiencies, hemoglobinopathies, and leukodystrophies.
Recent commercialization of lentiviral vector (LV)-based cell therapies and successful reports of clinical studies have demonstrated the untapped potential of LVs to treat diseases and benefit patients. Here, we report a method that solves all these problems for generation of stable LV packaging and producer cell lines exemplified by the construction of WinPac cells.There are two main strategies for producing LV in mammalian cells: transient transfection and stable transfection. Transient transfection introduces the nucleic acid for a limited time and does not integrate into the genome.
Lentiviral vectors (LVs) are potent tools for the delivery of genes of interest into mammalian cells and are now commonly utilised within the growing field of cell and gene therapy for the treatment of monogenic diseases and adoptive therapies such as chimeric antigen T-cell (CAR-T) therapy. Lentiviral vectors (LVs) efficiently can transduce both dividing and non-dividing cells and mediate transgene integration into the target cell genome, providing stable transgene expression. 1 Their versatility is further enhanced by the ability to pseudotype them with various heterologous envelope glycoproteins, enabling the transduction of a wi.The development of Good Manufacturing Practice (GMP) compliant instruments, technologies and techniques are essential for the development of LV-engineered T cell therapies. In this section we will present some applications of LV vector technologies for T cell engineering. Lentiviral vectors (LVs) are increasingly employed in gene and cell therapy. Standard laboratory production of LVs is not easily scalable, and research-grade LVs often contain contaminants that can interfere with downstream applications.
Lentivirus (LV) is an enveloped RNA virus that is characterized by its ability to incorporate viral RNA into host cell DNA. For this reason, LV vectors (LVV) are used in the cell and gene therapy field for delivery of nucleic acids into target cells both ex vivo and in vivo. Lentiviral vectors (LVs) are remarkable tools for gene transfer, both in vivo and in vitro, due to their ability to permanently integrate into the cell genome of dividing and non-dividing. Lentiviral vectors (LVs) are one of the most used transfer vehicles to deliver genetic material and the vector of choice to modify hematopoietic cells to correct primary immunodeficiencies, hemoglobinopathies, and leukodystrophies. Recent commercialization of lentiviral vector (LV)-based cell therapies and successful reports of clinical studies have demonstrated the untapped potential of LVs to treat diseases and benefit patients.
lentivirus cell line production
lentivirus cell line development pdf
lentivirus cell line conversion
chanel chance eau fraîche sephora
Very few databases containing information about shot scale do exist. Several pioneering computational works rely on the Cinemetrics.lv database to scrutinize the history of cinematic phenomena such as cut frequency in editing and shot scale (see for example ), examining how they vary across genres, historical patterns, etc. However, .
lv cell|lentivirus cell line workflow